Banded Sunflower Moth (E823, Revised August 2019)
Description
The banded sunflower moth, Cochylis hospes Walsingham, is a small (1/4 inch long), tan moth with a wingspan of about 1/2 inch. Its forewings have a triangular, dark brown band crossing through the middle of the wing (Figure 1). The peak of the triangle is oriented toward the leading margin of the wing. A smaller and less well-defined brown area is near the wing tips. The hind wing is light gray-brown and has no distinctive markings.
Banded sunflower moth eggs are small (approximately 1/50 inch diameter), round and appear white or opaque (Figure 1). Newly hatched larvae are off-white and about 1/16 inch long. The head capsule is dark brown, almost black. Larger, more mature larvae are red or green and about 1/2 inch long (Figure 1).
Life Cycle
Banded sunflower moths have one generation in North Dakota and the upper Great Plains. More than one generation is possible in the southern regions. Adults emerge about mid-July and are present in the field until mid-August. An individual moth lives between seven and 10 days, but moths are present for a total of about eight weeks each season because of the extended emergence period. Although some moths rest in the sunflower field during the day, many are found in vegetation (especially broadleaf vegetation) along field margins. At twilight, females move into the field to lay eggs.
Within a week after emergence, the moths begin to lay eggs on the bracts of the sunflower heads. Females preferentially deposit more eggs on midsized buds (R3, Figure 2) than smaller or larger buds. Very few eggs are laid on plants at pollen shed (R5.1, Figure 3) and later. Most eggs are laid singly or in small clusters, and are deposited on the outer whorl of bracts (Figure 1). A few eggs are placed on the inner bracts and the underside of the sunflower head. Eggs are present through early August and hatch about five to eight days after being deposited.
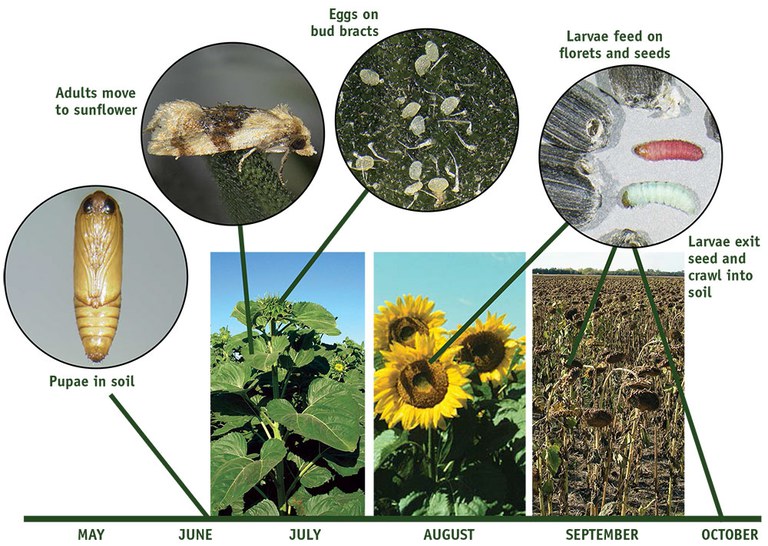
Figure 1. Life cycle of Cochylis hospes (banded sunflower moth).

Figure 2. Side view (left) and top view (right) of sunflower head at development stage R3.

Figure 3. Sunflower head at development stage R5.1.
Larvae develop through five instars, or stages, in about 10 to 14 days and are present in sunflower heads from mid-July to mid-September. Newly emerged larvae initially are found on and between the bracts and later move to the disk flowers and feed on pollen. Third and later instars tunnel through the disk flowers and feed on young, developing seeds. Each larva penetrates and consumes the contents of several seeds. After feeding to maturity, larvae drop to the ground and spin cocoons in the soil, where they pass the winter. The silken cocoons become coated with soil and are difficult to detect. Pupation takes place in late June or early July the following year. The pupal period lasts about 12 days.
Crop Damage
Larvae feeding on florets may prevent pollination and reduce the number of seeds the sunflower head produces. Once the larvae reach the third instar, they feed on seeds, usually consuming the entire kernel. The larva typically enters near the top of the seed and leaves by way of the same puncture after eating the kernel. Each larva typically consumes the kernels of six to seven seeds in addition to the early feeding that reduces seed number. Small areas of silken webbing on mature sunflower heads are indicators of banded sunflower moth larvae feeding in the seeds.
Individual seeds damaged by the banded sunflower moth larvae resemble those fed on by red sunflower seed weevil. However, the banded sunflower moth normally consumes the entire kernel, whereas the seed weevil larva consumes only about 1/3 of the kernel. Also, the entry-exit hole in the seed the banded sunflower moth creates is slightly larger than the one the seed weevil larva makes and usually is on the top rather than the side of the seed.
In most cases, banded sunflower moth larvae will have exited the seeds by the time the heads are harvested. In contrast, the red seed weevil larvae still may be in the seed at harvest and can cause heating and moisture problems in storage.
Distribution
The banded sunflower moth has been found from the Canadian Prairie Provinces of Saskatchewan and Manitoba to Texas and from the Dakotas and Colorado to New Jersey and North Carolina. The banded sunflower moth probably is present in all of the eastern United States where wild sunflowers are present. Nine species of wild sunflowers are known hosts.
Economic infestations of the banded sunflower moth are common in North Dakota. A related species, Cochylis arthuri Dang, has been reported infesting sunflowers in Saskatchewan and is found in small numbers in North Dakota. Cochylis arthuri has comparable feeding habits and development, and causes damage similar to that of the banded sunflower moth. Distinguishing C. arthuri from the banded sunflower moth is not necessary for pest management purposes.
Field Monitoring and Estimating Damage Potential
The use of insecticides to manage the banded sunflower moth is warranted when populations are at or exceed the economic injury level (EIL). The EIL is the density or number of insects expected to cause damage that is equal in value to the cost of control. Two distinct and separate procedures can be used to develop an EIL for the banded sunflower moth. The first samples for eggs and the second samples for the adult (moth) stage.
The egg sampling procedure is simpler and quicker than sampling for adult moths, and may provide more time to react if a treatment is needed. If the correct time to sample for eggs is missed, the adult moth sampling procedure should be used to determine the potential for economic damage.
Procedure 1. Egg Sampling
The potential for banded sunflower moth damage is determined by counting eggs on the outer layer of floral bracts in the field (Figure 4). Because the eggs are very small, you need a magnifier to count the small eggs accurately. We recommend using a head-mounted 3.5X magnifier (Figure 4) to leave both hands free for manipulating the bud being observed. Head-mounted magnifiers are available from most IPM suppliers. Egg counts should be made when most of the plants in the field are at plant stage R3 (distinct bud elongated ¾ inch above the nearest leaf, yellow ray petals not visible, see Figure 2). However, to avoid sampling bias, buds should be selected randomly without regard to plant stage.

Figure 4. Sampling for banded sunflower moth eggs using a head-mounted magnifier.
Sampling Steps
1) Divide each side of the field into two sections
2) Sample the center of each section at 20 feet into the field from the field edge
3) Randomly select five buds.
4) From each bud, randomly select six bracts from the outer whorl and count the eggs on each bract (Figure 5).
5) Average the egg counts from the five buds and then map the average egg counts from each sample site to a diagram of the field.
Next, compare the average egg density at each sampling site to the calculated economic injury level.
Economic Injury Level. The economic injury level (EIL, formula 1) is the number of eggs per six bracts and considers treatment cost ($/acre), market price ($/pound) and plant population per acre.
Economic Distance (ED). Because the numbers of banded sunflower moths often are higher on field margins (edge effect), and because this procedure only requires sampling eggs from the field margins, the data for each site pertains only to the area immediately around that location. If the average egg count at a sample point equals or exceeds the EIL, a second calculation is needed to estimate the population size further into the field (away from margin and toward the center of the field). Economic distance gives you the capability to diagram the extent of the EIL throughout the field (Figure 6) and is calculated by the following formula:
Individual ED values are calculated for each sample site around the field. ED measures the extent of the EIL from the sample site into the field and is given as distance from the field edge.
The formula for ED includes the value of the EIL (calculated using formula 1). D is the average egg density at 20 feet and e is the natural logarithm. Separate ED values are calculated for each sample site.
Instead of manually calculating the egg EIL and ED values, an ED Calculator is available to download from the NDSU Extension Service Entomology Web site under Entomology Updates and Sunflower section (www.ag.ndsu.nodak.edu/aginfo/entomology/entupdates/index.htm). The calculator will perform the calculations and provide a field diagram of the ED.

Figure 5. Sunflower bud showing a possible arrangement of six bracts (one bract outlined in red) to sample for banded sunflower moth eggs.
Example
Given a treatment cost of $8/acre, a market price of $0.15/pound and a plant population of 18,000/acre, the EIL would be 3.8 eggs per six bracts. Table 1 shows an example of egg counts at eight sample sites and the corresponding ED values. Figure 6 is a plot of the results. In this example, only a portion of the field (about 50 percent) would need a treatment (Figure 6).
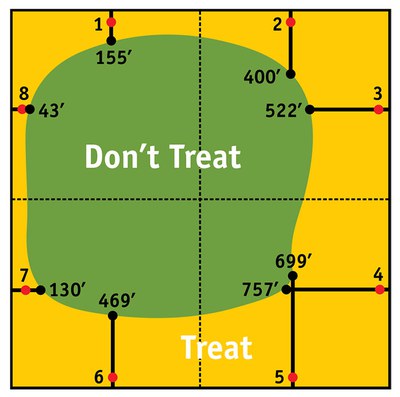
Figure 6. Field diagram showing economic distance (ED) and treat/no treat area of a field based on the data in Table 1. (Red dots indicate sampling sites.)
Procedure 2. Adult Moth Sampling
Sampling sites should be at least 75 to 100 feet from the field margins. In monitoring a field, use the X pattern (Figure 7), counting moths on 20 plants per sampling site to obtain the number of moths per 100 plants. Sampling should be conducted in the late bud stage (R3), usually during mid-July.
During the day (late morning or early afternoon), the moths remain quiet, resting on upper or lower surfaces of the leaves of sunflower plants. When disturbed, they flutter from plant to plant. When sampling for moths during day, the decision to treat or not is based on comparing the mean number of adult moths in the field to the EIL for moths. The EIL is the number of moths per head that will, if not managed, result in seed damage with a value equal to the cost of treatment. Use the following formula based on treatment costs, plant population and market price to determine the adult EIL for day sampling.
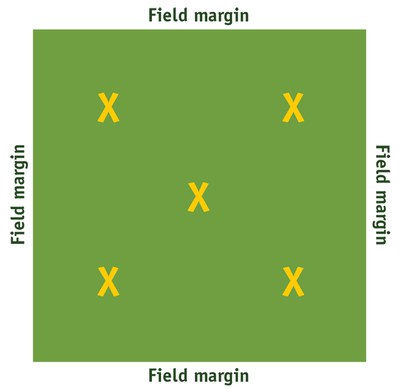
Figure 7. Sampling sites (X) for monitoring adult moths.
EIL for Adult Moth Sampling during the Day:
The constants in the formula simplify the calculation and include the amount of loss attributable to each banded sunflower moth larva produced per moth.
A sample calculation of the EIL based on moth sampling for the following conditions is given below.
Insecticide treatment cost = $8/acre
Market price = $0.15/pound
Plant population = 18,000/acre
For this set of variables, an infestation of about 1.0 moths per 100 plants will result in sufficient larvae to destroy seeds in the sunflower head equal to the $8 treatment cost per acre in a field of 18,000 plants per acre with a market value of 15 cents per pound. If the adult population has reached or exceeded this level, then the grower should consider the use of a chemical insecticide to prevent larval seed damage.
Instead of manually calculating the EIL, Tables 2 and 3 can be used to determine the EIL for a variety of market prices and for treatment costs of either $8 or $10 per acre.
Pheromone Trap Monitoring
A pheromone that attracts male banded sunflower moths is available commercially and can be placed in various types of traps for monitoring the presence of adults. However, research shows that using trap catches is not a reliable way to determine treatment thresholds. Therefore, pheromone trapping of moths should be used only to determine whether moths are emerging and present in the area.
Chemical Control and Application Timing
Any insecticide treatment must be applied at the appropriate plant growth stage of the sunflowers if egg moth sampling is used.
Although sampling is conducted for eggs, treatment is directed at the larval stage of the banded sunflower moth, which is the actual damaging stage. Once the decision to treat has been made, correctly timing the spray application to get maximum control is critical. The best sunflower plant stage to treat is the R5.1 growth stage (Figure 3), or when pollen shed is just beginning. This is the time when most banded sunflower moth eggs have hatched and larvae are present, but before the head has seeds forming. At this time, the larvae are beginning to feed on the disk flowers, are exposed on the head and are susceptible to the insecticide treatment. Application at an earlier growth stage may be warranted if monitoring reveals earlier than normal egg-laying activity. On older plants where the seeds have started maturing, most larvae will be feeding in the seeds or underneath the florets, and will be protected from the insecticide. By then, much of the feeding damage already has occurred.
Insecticides should be applied early in the morning or late in the day to minimize the adverse effect of the chemical on bees and other pollinators. For a list of insecticides registered for banded sunflower moth control in North Dakota, consult the “North Dakota Field Crop Insect Management Guide” Web site: www.ag.ndsu.edu/publications/crops/north-dakota-field-crop-insect-management-guide.
Cultural Control
Cultural control methods, such as tillage and planting date, have been effective in reducing damage from the banded sunflower moth. Deep fall plowing of sunflower stalks in Manitoba reduced moth emergence the following season by about 80 percent. However, unless this is done on an area-wide basis, the effect will be minimal. Research in North Dakota has demonstrated that delaying the planting of sunflowers until late May or early June will help reduce infestation levels of the banded sunflower moth. However, late planting may increase damage from the red sunflower seed weevil. Growers attempting to use planting dates to control any sunflower insect should be aware of all the insect pests they are likely to have, not just one species. They also should consider the effect of planting date on yield potential.
Biological Control
Beneficial insects, such as natural enemies and pollinators, should be conserved through the use of judicious management interventions. Predators and parasites can limit damage and help keep populations of the banded sunflower moth low, and pollinators can increase yield. Indiscriminant use of insecticides can reduce the efficacy of these beneficial insects.
Host Plant Resistance
Though wild, native sunflowers are often considered less susceptible to insect pests, some cultivated sunflower germplasm appears to have significant resistance to banded sunflower moth. The mechanisms of resistance in those lines is not clear, but the lines are part of ongoing efforts to understand how plant breeding can limit losses from this pest. Some commercial oilseed hybrids tested in multi-year field trials show very low susceptibility to the banded sunflower moth. Growers of such hybrids may be able prioritize other pests (such as the red sunflower seed weevil) or other management considerations over banded sunflower moth management.
This publication was authored by Janet Knodel, Extension entomologist and associate professor of entomology, and Dr. Laurence D. Charlet, USDA, ARS, Research Entomologist, NDSU, 2006.
Printing of this publication is supported by a Great Plains Diagnostic Network grant from a subcontract under the U.S. Department of Agriculture–National Institute of Food and Agriculture.
E823 (Revised August 2019)
North Dakota State University Fargo, North Dakota
NDSU Extension does not endorse commercial products or companies even though reference may be made to tradenames, trademarks or service names. NDSU encourages you to use and share this content, but please do so under the conditions of our Creative Commons license. You may copy, distribute, transmit and adapt this work as long as you give full attribution, don’t use the work for commercial purposes and share your resulting work similarly. For more information, visit www.ag.ndsu.edu/agcomm/creative-commons.
For more information on this and other topics, see www.ndsu.edu/extension
County commissions, North Dakota State University and U.S. Department of Agriculture cooperating. NDSU does not discriminate in its programs and activities on the basis of age, color, gender expression/identity, genetic information, marital status, national origin, participation in lawful off-campus activity, physical or mental disability, pregnancy, public assistance status, race, religion, sex, sexual orientation, spousal relationship to current employee, or veteran status, as applicable. Direct inquiries to Vice Provost for Title IX/ADA Coordinator, Old Main 201, NDSU Main Campus, 701-231-7708, ndsu.eoaa@ndsu.edu. This publication will be made available in alternative formats for people with disabilities upon request, 701-231-7881. 1M-11-06, 3M-5-10, 2M-8-19

